The Hidden Brain Switch That Tells You to Stop Eating: What the New Astrocyte Research Means
The Short Version
- A study published in PNAS on April 6, 2026 found that astrocytes — long considered mere support cells — actively relay the fullness signal in your brain via a previously unknown pathway.
- The chain runs from tanycytes (glucose sensors) to lactate to astrocytes to glutamate to POMC neurons — the brain's 'stop eating' cells — and takes time to complete.
- The astrocyte/HCAR1 pathway is entirely separate from GLP-1, meaning it could eventually complement drugs like Ozempic rather than compete with them.
- The signal chain has latency, which is one reason eating slowly supports better portion regulation — your brain needs time to catch up.
- This research reframes overeating as a biology and timing issue, not a willpower failure — your body's relay system was designed for a different pace than modern eating demands.
The Question Nobody Had Fully Answered

The Question Nobody Had Fully Answered
You finish a meal and at some point, you stop eating. It feels obvious, almost automatic — your body just knows. But the mechanism behind that knowing has been surprisingly hard to pin down. Scientists understood pieces of it: hormones like leptin and GLP-1 send signals, neurons in the hypothalamus respond, and somewhere in that chain the message "enough" gets transmitted. What they didn't fully understand was how the brain translates rising blood sugar into an actual stop signal.
A study published in the Proceedings of the National Academy of Sciences on April 6, 2026 offers a significant new piece of that puzzle — and the answer involves a type of brain cell that researchers had long assumed was just background support.

The research, led by scientists at the University of Concepción in Chile in collaboration with the University of Maryland, identified a previously unknown communication chain inside the hypothalamus — the brain region responsible for regulating hunger and fullness. At the center of it are cells called astrocytes. And what they found changes how we understand the moment your brain decides you've had enough.
The Unexpected Middleman

The Unexpected Middleman
Astrocytes are among the most abundant cells in the brain. For decades, scientists classified them as support staff — essential for neuronal function, but not direct players in signaling. The neurons were the actors; the astrocytes kept the stage in order.
That assumption is now significantly complicated.
The new PNAS study found that astrocytes aren't passive bystanders in appetite regulation — they're active relays in a previously unrecognized communication chain. And the signal that activates them isn't a hormone or a nerve impulse in the traditional sense. It's lactate.
"Researchers used to think that lactate produced from tanycytes 'spoke' directly to neurons involved in appetite control. But we found that there was an unexpected middleman in that conversation — astrocytes."
— Ricardo Araneda, University of Maryland
The middleman metaphor is apt. Astrocytes sit between the cells that detect rising glucose and the neurons that actually fire the "stop eating" signal. Without them, the message doesn't get through.
How the Signal Works

How the Signal Works
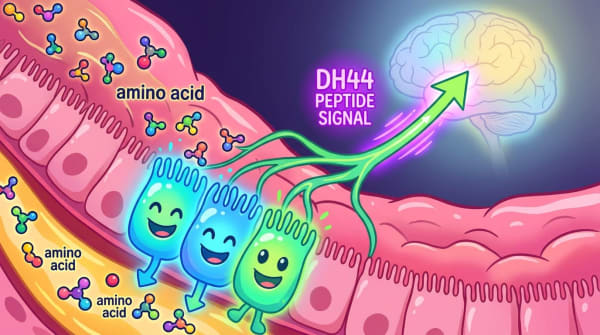
The communication chain begins with specialized cells called tanycytes, which line a fluid-filled cavity deep inside the brain and act as glucose sensors. When blood sugar rises after a meal, tanycytes process that sugar and release a byproduct called lactate into the surrounding brain tissue. That lactate then reaches neighboring astrocytes.
Here's where the new finding comes in: astrocytes carry a receptor called HCAR1 that detects lactate. When lactate binds to HCAR1, astrocytes activate and release glutamate — a neurotransmitter that then stimulates POMC neurons, the brain's primary "stop eating" cells.
The chain looks like this:
What makes this doubly interesting is the dual effect the researchers observed. The hypothalamus contains two opposing populations of neurons — those that promote hunger and those that suppress it. As ScienceDaily reported, the lactate signal may work on both simultaneously: activating the fullness neurons through astrocytes while potentially quieting the hunger neurons through a more direct route. It's like hitting the brakes on hunger from two directions at once.
One experiment illustrated the network's sensitivity: researchers delivered glucose into a single tanycyte while monitoring surrounding astrocytes. That one cell's activity was enough to trigger responses in multiple neighboring astrocytes — a tiny, localized metabolic event rippling outward through the brain's network.
What This Means for Ozempic and Beyond

What This Means for Ozempic and Beyond
The timing of this research matters. We are living through a cultural moment shaped by GLP-1 receptor agonists — Ozempic, Wegovy, Mounjaro — drugs that work by mimicking the hormone GLP-1 to suppress appetite and slow digestion. They work remarkably well for many people. They are also expensive, not universally accessible, and come with side effects that make them unsuitable for others.
The astrocyte pathway is completely separate from the GLP-1 system. It operates through a different mechanism, in different cells, via a different receptor. That's significant — not because it replaces GLP-1 drugs, but because it suggests a complementary target.
"We now have a different mechanism where we might be able to target astrocytes or specifically this HCAR1 receptor," lead researcher Araneda told ScienceDaily. "It would be a novel target that may complement existing therapies like Ozempic, for example, and improve the lives of many who suffer from obesity and other appetite-related conditions."
It's worth noting that this study was conducted in animal models. Tanycytes and astrocytes are present in all mammals, including humans, and the researchers' next steps involve investigating whether manipulating the HCAR1 receptor directly can change feeding behavior — a necessary step before any clinical application. This is promising early-stage science, not an imminent treatment.
But the direction is clear: the brain's appetite regulation system is more complex, and more targetable, than we knew.
What It Means for You Right Now

What It Means for You Right Now
The honest answer is: probably not much changes for your next meal. This research isn't a supplement you can take or a behavior you can immediately adjust. What it offers is something more foundational — a clearer picture of what your body is actually doing when it's working correctly.
Your brain is not simply waiting for your stomach to stretch. It's running a sophisticated chemical relay — glucose to lactate to astrocytes to glutamate to neurons — that takes time to complete. That has real practical implications.
It's one reason eating slowly tends to support better portion regulation. The signal chain has latency. Give it time to fire before deciding you need more. It's also one reason that highly processed foods, engineered to override satiety signals, are so effective at doing exactly that — they short-circuit a system that was designed for a different pace of eating.
What would it mean to eat in a way that actually works with this system — to slow down enough to let your own brain catch up?
Understanding how fullness actually works doesn't automatically make eating easier. But it does change the conversation from willpower and discipline to biology and timing. You are not failing some test of character when you eat past fullness. You may simply be moving faster than your brain's relay system can keep up.
The science of why you stop eating is, it turns out, far richer than anyone assumed. Astrocytes — the quiet ones, the support staff — are doing more than anyone gave them credit for.
Content ID: gLBAvx4OWHi5ocujnkzOGBMr